Biomedical Engineering Staffing Agency
Nationwide biomedical engineering recruitment for medical device, pharmaceutical, biotech, diagnostics, and research organizations. Contract, contract-to-hire, and direct hire solutions.

Biomedical Engineering Talent That Understands Regulated Environments.
If you’ve tried hiring biomedical engineers before, you already know the problem. Resumes look strong. Degrees check out. But once interviews start, the gaps show up — no real validation ownership, no exposure to FDA design controls, no experience inside GMP production, and strong R&D capability with limited documentation discipline.
Biomedical engineering sits in a narrow lane between engineering depth and healthcare regulation. That intersection is where hiring mistakes get expensive. KORE1 supports biomedical engineering hiring nationwide across medical device manufacturers, pharmaceutical production environments, biotech labs, diagnostics companies, research institutions, and healthcare technology teams. Biomedical engineering is a specialized vertical within our broader engineering staffing agency practice, where technical discipline and regulatory environment knowledge matter most. Companies building cross-functional device teams often also need engineers from our mechanical engineering staffing practice for enclosure design, thermal management, and manufacturing-ready prototyping, and our electrical engineering staffing team for circuit design, embedded firmware, and power systems work.
What Is a Biomedical Engineering Staffing Agency?
It’s not general engineering recruiting. A biomedical engineering staffing agency hires engineers who work inside healthcare-driven environments — places where documentation matters, quality systems exist, and product decisions can carry regulatory weight.
That might mean building a Class II medical device, qualifying equipment on a pharmaceutical production line, writing validation protocols in a biotech lab, or integrating diagnostic systems into hospital infrastructure. The work changes. The environment is what ties it together.
Biomedical engineering is formally described by the National Institute of Biomedical Imaging and Bioengineering as a field that blends engineering and medical science, and is categorized as its own occupation by the U.S. Bureau of Labor Statistics. The National Science Foundation reports U.S. R&D spending above $800 billion annually, with life sciences representing a significant share.

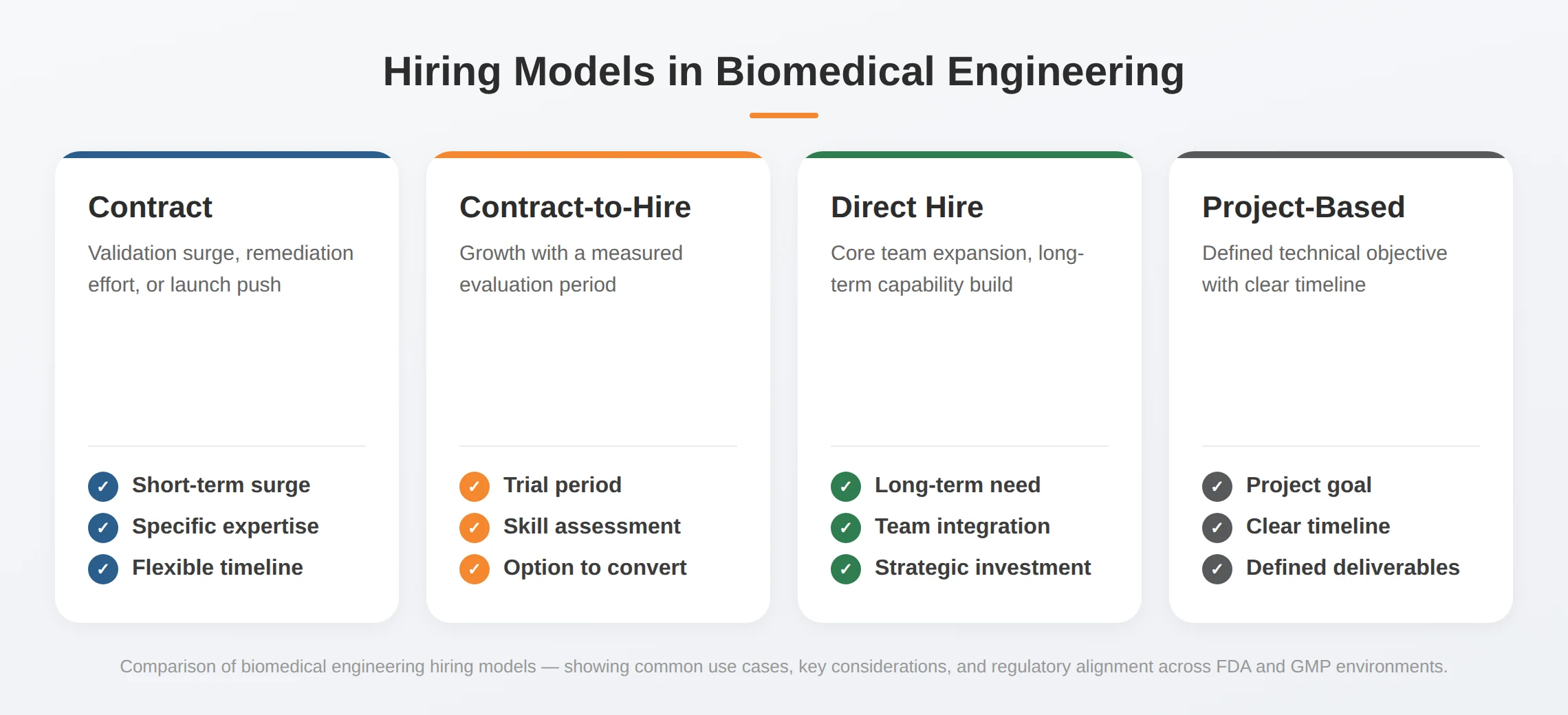
Flexible Engagement Models for Biomedical Engineering
Whether you need short-term validation support or a permanent product development team, we match the hiring model to your product stage and timeline.
Where Biomedical Engineers Actually Contribute
The title covers more ground than most job descriptions suggest
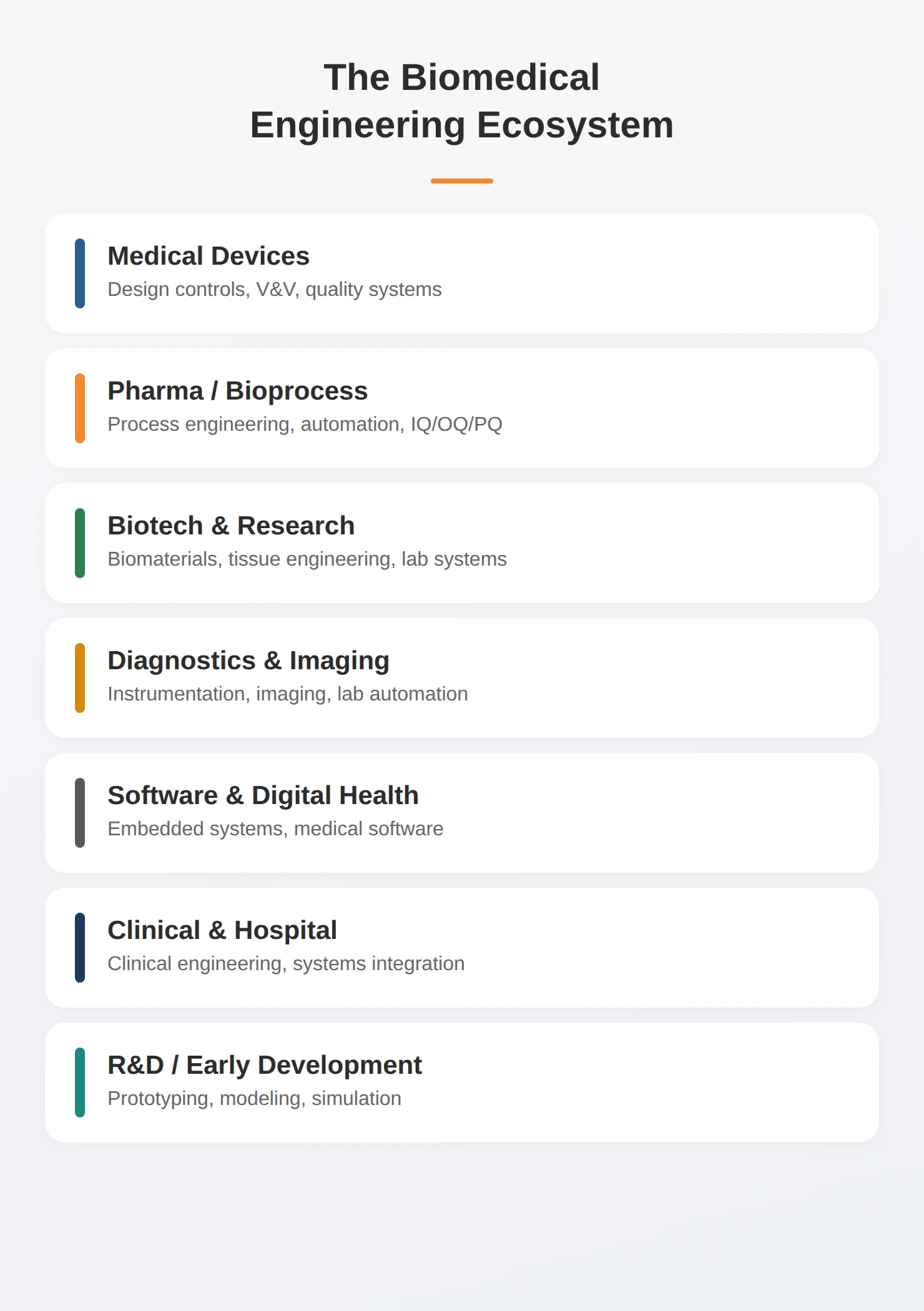
The title “biomedical engineer” covers more ground than most job descriptions suggest. Some are building early prototypes. Some are writing verification protocols. Some are qualifying production equipment. Some are troubleshooting hospital systems. Hiring needs commonly span:
- Product design and development
- Systems engineering
- Embedded and firmware development
- Manufacturing and automation
- Process validation
- Quality systems
- Risk management
- Clinical systems integration
That spread is why generalized recruiting approaches fall short.

Medical Device Engineering
Device teams often need engineers who understand design controls, risk analysis (ISO 14971), design history files, verification & validation strategy, and post-market change management.
These environments operate under 21 CFR Part 820 and ISO 13485 frameworks enforced in the United States by the U.S. Food and Drug Administration. Experience here is specific, procedural, and shows up quickly in interviews.
Pharmaceutical and Bioprocess Environments
Biomedical engineers in pharma settings are frequently involved in manufacturing systems, equipment qualification (IQ/OQ/PQ), process engineering, automation and controls, and GMP documentation.
Pharmaceutical production follows current Good Manufacturing Practices under 21 CFR Parts 210 and 211. In these environments, engineers need to understand deviation handling, change control, documentation flow, and validation reporting — not just technical theory.
Biotech, Diagnostics, and Clinical Systems
Biotech & Translational Research
Biomaterials development, tissue engineering, laboratory systems, and clinical manufacturing support.
Diagnostics & Imaging
Instrumentation systems, imaging hardware, and laboratory automation requiring regulated environment experience.
Clinical Engineering
Hospital-based biomedical equipment management, systems integration, and device lifecycle oversight.
The Labor Market Context
Biomedical engineering is a defined occupation category. The U.S. Bureau of Labor Statistics reports a median annual wage of approximately $99,550 (May 2023).
| Hiring Model | Common Scenario |
|---|---|
| Contract | Validation surge, remediation effort, launch push |
| Contract-to-Hire | Growth with measured evaluation period |
| Direct Hire | Core team expansion or long-term capability build |
| Project-Based | Defined technical objective with timeline |
Competition is typically strongest for engineers with hands-on regulatory exposure, validation ownership, and manufacturing systems experience. The model should match product stage, compliance exposure, timeline pressure, and internal capacity.
How We Approach Biomedical Engineering Searches
Biomedical engineering searches require clarity upfront. Before sourcing begins, we identify the full picture.
Define the Environment
Product lifecycle stage, regulatory environment, validation ownership expectations, manufacturing exposure requirements, and software versus hardware emphasis.
Evaluate Project History
We assess candidates based on what they built, what they validated, what systems they worked inside, and what documentation they owned — not just what’s listed.
Deliver and Support
We present qualified candidates who match the technical depth and regulatory awareness your team requires, with ongoing support through placement.
Industries We Support
- Medical device manufacturers
- Pharmaceutical producers
- Biotech companies
- Diagnostic and imaging firms
- Research institutions
- Healthcare technology teams
- Contract manufacturing organizations
- Hospitals and clinical systems
Ready to Strengthen Your Biomedical Engineering Team?
Whether you’re expanding engineering capacity, preparing for inspection, strengthening validation ownership, or scaling production environments, we can support the search with focus and technical precision.
Frequently Asked Questions
What does a biomedical engineering staffing agency actually handle?
We recruit engineers who work on healthcare technologies, including device development, validation work, pharmaceutical production systems, diagnostics equipment, and clinical infrastructure. The focus isn’t just technical skill — it’s whether the engineer understands the operating environment.
How long does hiring usually take?
It depends on what you’re asking for. A contract validation engineer tied to a short-term push can move quickly. A direct hire requiring regulatory ownership or systems depth will typically take longer to identify and vet.
Is regulatory background necessary for biomedical engineers?
In many biomedical environments, yes. Device and pharmaceutical settings operate within structured quality systems. Engineers who have worked inside those systems tend to ramp faster, navigate documentation requirements more effectively, and reduce compliance risk.
What education do most biomedical engineers have?
Most hold a bachelor’s degree in biomedical engineering or a related field. Beyond that, employers often look for experience in validation, manufacturing systems, design controls, and cross-functional development.
Is contract staffing common in biomedical engineering?
Very common. Contract engineers are frequently engaged during validation cycles, inspection preparation, product launch timelines, research surges, and remediation efforts.
